
A case report of misdiagnosed adenoid cystic carcinoma of the parotid gland on ultrasound: imaging-pathological correlation and diagnostic implications
Highlight box
Key findings
• This case reports a misdiagnosed adenoid cystic carcinoma (ACC) of the parotid gland in a 46-year-old female, initially misinterpreted as a pleomorphic adenoma on ultrasound due to “benign-mimicking” features (well-defined margins, homogeneous hypoechogenicity, posterior acoustic enhancement, and sparse vascularity). Definitive diagnosis required postoperative pathology (cribriform/trabecular patterns; SOX10/p63-positive; Ki-67 3%; MYB::NFIB fusion-negative), highlighting ACC’s heterogeneous imaging presentation as a key diagnostic pitfall.
What is known and what is new?
• ACC frequently mimics benign tumors on conventional ultrasound, leading to preoperative misdiagnosis. Its aggressive behavior (perineural invasion, delayed metastasis) contrasts with indolent growth.
• This case illustrates the potential diagnostic value of integrating multimodal imaging (elastography for tumor stiffness, contrast-enhanced ultrasound (CEUS) for washout patterns, computed tomography/magnetic resonance imaging for detecting suspicious perineural spread) with molecular biomarkers. Furthermore, in this case, the observed “benign-mimicking” sonographic features were associated with a low Ki-67 proliferation index (3%), which may suggest a potential link between proliferative inactivity and a less aggressive imaging phenotype.
What is the implication, and what should change now?
• Clinicians should suspect ACC in solid parotid tumors with deceptively benign ultrasound features, particularly when elastography shows high stiffness or CEUS reveals malignant washout. Ultrasound-guided core needle biopsy is recommended for equivocal cases. Based on the challenges highlighted in this case, further research could be directed toward exploring multimodal diagnostic models that combine imaging parameters with molecular biomarkers. Such efforts may help elucidate the complex relationship between pathological features and imaging presentations in ACC.
Introduction
Adenoid cystic carcinoma (ACC) is a rare malignant neoplasm and one of the primary malignant salivary gland tumors (1). Although characterized by indolent growth, ACC exhibits aggressive biological behaviors, including a propensity for perineural invasion and delayed distant metastasis (2). The 5-year survival rate may reach 85%, yet the long-term prognosis deteriorates markedly, with survival rates declining to approximately 30% (1). Imaging serves as a cornerstone for preoperative diagnosis and staging evaluation, playing a pivotal role in surgical planning and prognostic prediction. However, the diagnostic efficacy of a single imaging modality is frequently limited by the heterogeneous imaging manifestations of ACC (3). Currently, the clinical value of multimodal imaging integration in ACC diagnosis remains understudied. Emerging techniques such as elastography and contrast-enhanced ultrasound (CEUS) may quantitatively assess tumor stiffness and microvascular characteristics (4,5), while computed tomography (CT) and magnetic resonance imaging (MRI) can compensate for ultrasonography’s limitations in evaluating deep anatomical structures. Through a misdiagnosed case of parotid ACC, this study systematically analyzes its imaging-pathological correlation and proposes a multimodal diagnostic strategy to enhance preoperative diagnostic accuracy. We present this article in accordance with the CARE reporting checklist (available at https://gs.amegroups.com/article/view/10.21037/gs-2025-354/rc).
Case presentation
Clinical history
A 46-year-old female presented with a painless mass in the left cheek persisting for over one year, with transient enlargement of the mass following spicy food intake and spontaneous resolution. No associated symptoms, such as pain, skin ulceration, or pharyngeal foreign body sensation, were noted. Physical examination revealed a firm, mobile mass, approximately 3.0 cm × 2.0 cm by palpation, in the left parotid region, with mild tenderness.
Imaging findings
Ultrasonography
Gray-scale ultrasound identified a well-defined, regular-shaped hypoechoic mass (2.5 cm × 2.0 cm × 2.0 cm) within the left parotid gland. The lesion demonstrated homogeneous internal echogenicity without calcification or cystic components, accompanied by posterior acoustic enhancement (Figure 1A). Color Doppler flow imaging (CDFI) revealed punctate/linear vascular signals within the mass (Figure 1B), suggestive of pleomorphic adenoma.

Enhanced CT
A soft tissue nodule (2.4 cm × 2.1 cm) with well-demarcated margins was observed in the left parotid gland. The mass was located anterosuperolateral to the retromandibular vein, with a clear boundary between the lesion and the vessel. The unenhanced CT attenuation was 30.23 HU. Following contrast administration, the lesion’s attenuation increased to 45 HU in the arterial phase (28 s), further rose to 89.43 HU in the venous phase (65 s), and showed persistent enhancement of 65 HU in the delayed phase. No adjacent tissue invasion or lymphadenopathy was identified (Figure 2).

Surgical and pathological findings
- Gross specimen: a well-circumscribed, encapsulated mass (2.0 cm × 2.0 cm × 2.0 cm) was identified in the superficial lobe of the left parotid gland, showing no adhesion to surrounding tissues.
- Histopathology: microscopic examination demonstrated nodular tumor tissue composed of bilateral epithelial cells (ductal and myoepithelial) arranged in cribriform, trabecular, and solid patterns. Necrosis and perineural invasion were absent.
- Immunohistochemistry: ductal epithelium: CK7(+), focal EMA(+), S100(−), SOX10(+).
- Myoepithelium: P63(+), SOX10(+), S100(−), EMA(−), SMA(+).
- Ki-67 proliferation index: 3% (Figure 3).
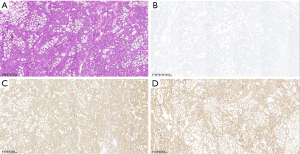 Figure 3 Histopathological and immunohistochemical findings of the parotid gland adenoid cystic carcinoma. (A) Hematoxylin and eosin staining (original magnification ×30) reveals characteristic tumor architectures, including cribriform and trabecular patterns. (B) Ki-67 immunohistochemical stain (original magnification ×35) shows a low proliferative index (approximately 3%), consistent with the tumor’s indolent behavior. (C) p63 immunohistochemical stain (original magnification ×25) demonstrates positive nuclear staining in myoepithelial cells, supporting the diagnosis. (D) SOX10 immunohistochemical stain (original magnification ×25) shows strong nuclear positivity in both ductal and myoepithelial components, confirming the diagnosis of adenoid cystic carcinoma.
Figure 3 Histopathological and immunohistochemical findings of the parotid gland adenoid cystic carcinoma. (A) Hematoxylin and eosin staining (original magnification ×30) reveals characteristic tumor architectures, including cribriform and trabecular patterns. (B) Ki-67 immunohistochemical stain (original magnification ×35) shows a low proliferative index (approximately 3%), consistent with the tumor’s indolent behavior. (C) p63 immunohistochemical stain (original magnification ×25) demonstrates positive nuclear staining in myoepithelial cells, supporting the diagnosis. (D) SOX10 immunohistochemical stain (original magnification ×25) shows strong nuclear positivity in both ductal and myoepithelial components, confirming the diagnosis of adenoid cystic carcinoma. - Final diagnosis: ACC.
- Molecular testing: fluorescence in situ hybridization (FISH) was negative for MYB::NFIB gene fusion.
Treatment and follow-up
Given the strong clinical and radiological suspicion for a benign tumor, a formal metastatic workup for staging was not performed preoperatively. The patient subsequently underwent complete resection of the tumor and the superficial lobe of the parotid gland with preservation of the facial nerve.
Postoperative pathological staging was conducted, with a particular focus on the presence or absence of pulmonary metastases—a critical concern in ACC management. The disease was confirmed to be stage pT2N0M0 according to the American Joint Committee on Cancer (AJCC) 8th edition criteria. Postoperative adjuvant radiotherapy was administered one month later. The follow-up strategy, with a particular focus on detecting pulmonary metastases, includes ultrasonographic surveillance and contrast-enhanced CT or MRI of the head, neck, and chest. To date, no evidence of local recurrence or distant metastasis has been identified.
Ethical approval and consent
All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration and its subsequent amendments. Written informed consent was obtained from the patient for the publication of this case report and accompanying images. A copy of the written consent is available for review by the editorial office of this journal.
Discussion
Imaging features and differential diagnosis of ACC
The biological characteristics of ACC in this case contribute to its frequent overlap in sonographic features with benign lesions. Unlike the classically infiltrative growth pattern typical of many malignancies, this case exhibited an expansile growth morphology. This pattern underlies its “benign-mimicking” sonographic presentation, which includes well-defined margins, regular morphology, homogeneous hypoechogenicity, and posterior acoustic enhancement.
The posterior acoustic enhancement, a finding more commonly associated with fluid-filled or homogeneous solid benign lesions, can be attributed to the tumor’s histologically uniform architecture and lack of sound-attenuating components. Specifically, the absence of calcifications, coarse fibrous septa, or other shadowing-inducing interfaces allows greater through-transmission of sound waves.
The homogeneous hypoechogenicity is directly correlated with its distinct histopathological features. The tumor is composed of ductal and myoepithelial cells organized in cribriform, trabecular, and solid patterns, forming numerous minute and acoustically uniform interfaces. Moreover, the lack of necrosis or cystic degeneration—which typically results in echogenic heterogeneity—is consistent with the tumor’s indolent biological behavior. This is further supported by the low Ki-67 proliferation index (3%), indicating minimal proliferative activity and reduced propensity for ischemia-induced necrosis commonly seen in rapidly growing tumors (6). The absence of other echogenicity-disrupting features, such as dystrophic calcifications or prominent fibrous bands, further reinforces the homogeneous sonographic appearance.
Histologically, ACC is characterized by tumor cells arranged in cribriform, tubular, or solid architectures (7), accompanied by abundant myoepithelial cells and collagen fibers, which confer increased tumor stiffness. This is reflected in ultrasound elastography as a higher strain ratio compared to benign counterparts (8).
Most benign parotid tumors (e.g., pleomorphic adenoma) demonstrate hypovascularity on CDFI, manifesting as sparse or absent intratumoral vascularity (9), consistent with their low proliferative activity and microvessel density. In contrast, malignancies such as mucoepidermoid carcinoma typically display hypervascularity, correlating with high proliferative activity and overexpression of angiogenic factors. However, in this case, ACC exhibited CDFI features resembling benign tumors, with only sparse punctate/linear vascular signals. The hypovascular nature of ACC may arise from its unique tumor microenvironment: despite localized expression of pro-angiogenic factors like vascular endothelial growth factor (VEGF), its vascular density remains significantly lower than that of other aggressive malignancies (10). Overreliance on CDFI morphological features alone may thus lead to misdiagnosis.
CEUS offers distinct advantages in ACC differentiation: quantitative hemodynamic parameters (e.g., peak intensity, time-to-peak, and area under the curve) enable objective assessment of tumor microcirculation (11). ACC frequently demonstrates an early-phase hyperenhancement with rapid washout on CEUS, whereas pleomorphic adenomas exhibit slow, homogeneous enhancement (12). Therefore, when conventional ultrasound findings are equivocal, integrating CEUS features with tumor microenvironment biomarker expression levels can provide critical diagnostic evidence for differentiating ACC from benign tumors.
Imaging features of CT and MRI in ACC diagnosis
CT typically reveals ACC as a well-defined soft tissue mass displaying heterogeneous enhancement in the arterial phase, likely attributable to uneven distribution of stromal components within the tumor. Persistent enhancement is observed in the delayed phase, with an increase in enhancement intensity compared to the arterial phase. Furthermore, the enhancement pattern correlates with tumor aggressiveness, with some cases demonstrating peripheral tissue infiltration or bony invasion (13).
MRI provides superior soft tissue contrast resolution, enabling precise delineation of tumor margins, internal architecture, and suspicious perineural spread. On T1WI, ACC typically exhibits iso- to hypointense signals, while appearing hyperintense on T2WI (14). Diffusion-weighted imaging and apparent diffusion coefficient (ADC) maps further aid in differentiating benign from malignant lesions, with ACC generally demonstrating lower ADC values reflecting high cellular density (15). Additionally, MRI serves as an important modality for evaluating radiological features suggestive of perineural spread, such as nerve thickening, enhancement, and foraminal widening. This assessment offers critical insights for preoperative tumor delineation, surgical planning, and prognostic evaluation.
Correlation between molecular pathology and imaging features
In this case, the Ki-67 proliferation index of 3% indicates low tumor proliferative activity, which may partially account for the “benign-mimicking” imaging features observed (16). This finding highlights the potential value of correlating immunohistochemical markers with imaging characteristics (e.g., enhancement patterns, margin definition) to improve diagnostic accuracy (17).
Multimodal imaging integration strategy
Ultrasound, CT, and MRI exhibit complementary diagnostic roles in ACC evaluation: ultrasound provides real-time dynamic assessment of tumor size, morphology, margin characteristics, and vascular distribution. CT, with its superior bone resolution, is indispensable for detecting calcifications and osseous destruction (18). MRI excels in soft tissue contrast resolution, delineating intratumoral heterogeneity, perineural invasion, and adjacent tissue infiltration (19). Using ultrasound as a preliminary screening method to quickly locate lesions, and then evaluate the extent of tumor invasion through CT/MRI, which can provide more information for accurate diagnosis and treatment planning of ACC by integrating multimodal imaging data.
Clinical decision-making and diagnostic pitfalls in context
The present case also offers valuable insights into clinical decision-making for parotid masses. Although current international guidelines, such as those from European Society for Medical Oncology (ESMO) and American Society of Clinical Oncology (ASCO), strongly recommend preoperative fine-needle aspiration biopsy (FNAB) to optimize surgical planning and avoid multiple procedures, this case illustrates a scenario where the standard pathway was not followed. The decision to proceed directly to surgery was based on a confluence of factors: the unequivocally benign appearance on both ultrasound and CT, which significantly lowered the pre-test probability of malignancy, and the patient’s informed preference for a single, definitive intervention after detailed counseling, which considered personal and socioeconomic circumstances within our local healthcare context.
Crucially, the fact that the preoperative findings were so convincingly “benign” that they justified a deviation from the standard biopsy pathway underscores the profound diagnostic challenge posed by ACC. This case serves as a powerful reminder that the imaging features of ACC can be deceptively benign, not merely at a morphological level but to the extent that they can influence the entire diagnostic workflow. Therefore, this report highlights a critical pitfall: an overreliance on seemingly benign imaging features can lead to a false sense of security, resulting in the omission of biopsy and potentially necessitating completion surgery after an unexpected diagnosis of malignancy. Our experience reinforces the importance of maintaining a high index of suspicion for ACC even in the face of classic benign features, particularly when patient and contextual factors favor a direct surgical approach.
For solid parotid tumors with well-defined margins, clinicians must maintain a high index of suspicion for ACC, particularly when ultrasound elastography reveals elevated stiffness values or CEUS demonstrates malignant enhancement patterns, even in the absence of typical infiltrative features. In such diagnostic dilemmas, an ultrasound-guided core needle biopsy is recommended to achieve precise histopathological confirmation and guide personalized therapeutic strategies.
Conclusions
This case underscores the diagnostic challenges of ACC, in which conventional ultrasound features significantly overlap with benign tumors. It further demonstrates how this overlap can potentially mislead clinical decision-making, bypassing standard diagnostic safeguards. Integrating elastography, CEUS, CT, MRI, and molecular biomarkers (e.g., Ki-67) could be a valuable strategy for improving the diagnostic accuracy of ACC in clinically ambiguous scenarios. Based on the challenges highlighted in this case, further research could be directed toward exploring multimodal diagnostic models that combine imaging parameters with molecular biomarkers. Such efforts may help elucidate the complex relationship between pathological features and imaging presentations in ACC.
Acknowledgments
None.
Footnote
Reporting Checklist: The authors have completed the CARE reporting checklist. Available at https://gs.amegroups.com/article/view/10.21037/gs-2025-354/rc
Peer Review File: Available at https://gs.amegroups.com/article/view/10.21037/gs-2025-354/prf
Funding: None.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://gs.amegroups.com/article/view/10.21037/gs-2025-354/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration and its subsequent amendments. Written informed consent was obtained from the patient for the publication of this case report and accompanying images. A copy of the written consent is available for review by the editorial office of this journal.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Zupancic M, Näsman A, Friesland S, et al. Adenoid Cystic Carcinoma, Clinical Presentation, Current Treatment and Approaches Towards Novel Therapies. Anticancer Res 2024;44:1325-34. [Crossref] [PubMed]
- Cantù G. Adenoid cystic carcinoma. An indolent but aggressive tumour. Part B: treatment and prognosis. Acta Otorhinolaryngol Ital 2021;41:296-307. [Crossref] [PubMed]
- Cavaliere M, De Luca P, Scarpa A, et al. Acinic cell carcinoma of the parotid gland: from pathogenesis to management: a literature review. Eur Arch Otorhinolaryngol 2020;277:2673-9. [Crossref] [PubMed]
- Dajti E, Huber AT, Ferraioli G, et al. Advances in imaging-Elastography. Hepatology 2025; Epub ahead of print. [Crossref]
- Fei X, Zhu L, Han P, et al. Value of high frame rate contrast-enhanced ultrasound in predicting microvascular invasion of hepatocellular carcinoma. Insights Imaging 2024;15:273. [Crossref] [PubMed]
- Karabulut E, Akbulut S, Samdanci ET, et al. Are Ki-67 and Procalcitonin Expression Levels Useful in Predicting the Biological Behavior of Hepatocellular Carcinoma After Liver Transplantation? J Clin Med 2024;14:144. [Crossref] [PubMed]
- Powell SK, Kulakova K, Kennedy S. A Review of the Molecular Landscape of Adenoid Cystic Carcinoma of the Lacrimal Gland. Int J Mol Sci 2023;24:13755. [Crossref] [PubMed]
- Shi H, Qiu W, Yang P, et al. Diagnosis of pulmonary adenoid cystic carcinoma using multimodal ultrasound imaging technology: A case report. Medicine (Baltimore) 2025;104:e41212. [Crossref] [PubMed]
- Zhao L, Mao Y, Mu J, et al. The diagnostic value of Superb Microvascular Imaging in identifying benign tumors of parotid gland. BMC Med Imaging 2020;20:107. [Crossref] [PubMed]
- Guldogan N, Esen G, Kayadibi Y, et al. Adenoid Cystic Carcinoma of the Breast: Multimodality Imaging Findings and Review of the Literature. Acad Radiol 2023;30:1107-17. [Crossref] [PubMed]
- Shi L, Wu D, Yang X, et al. Contrast-Enhanced Ultrasound and Strain Elastography for Differentiating Benign and Malignant Parotid Tumors. Ultraschall Med 2023;44:419-27. [Crossref] [PubMed]
- Peng L, Luo Y, Li N, et al. Value of contrast-enhanced ultrasound in differential diagnosis of salivary adenoid cystic carcinoma and pleomorphic adenoma. Academic Journal of Chinese Pla Medical School 2021;42:172-6.
- Ceylanoğlu KS, Konuk O. Clinical and radiologic outcomes of pleomorphic adenoma and adenoid cystic carcinoma of the lacrimal gland. Arq Bras Oftalmol 2023;86:359-64. [Crossref] [PubMed]
- Wang Y, Guo X, Yu K, et al. Adenoid cystic carcinoma of head and neck: Summary and review of imaging findings. Heliyon 2023;9:e21901. [Crossref] [PubMed]
- Orhan Soylemez UP, Atalay B. Differentiation of Benign and Malignant Parotid Gland Tumors with MRI and Diffusion Weighted Imaging. Medeni Med J 2021;36:138-45. [Crossref] [PubMed]
- Park S, Nam SJ, Keam B, et al. VEGF and Ki-67 Overexpression in Predicting Poor Overall Survival in Adenoid Cystic Carcinoma. Cancer Res Treat 2016;48:518-26. [Crossref] [PubMed]
- Zhang L, Xiao Y, Dong M, et al. Three-dimensional MR elastography-based stiffness for assessing the status of Ki67 proliferation index and Cytokeratin-19 in hepatocellular carcinoma. Eur Radiol 2025;35:4722-35. [Crossref] [PubMed]
- Ouatassi N, Elguerch W, Bensalah A, et al. Unusual presentation of parotid gland adenoid cystic carcinoma : A case presentation and literature review. Radiol Case Rep 2022;17:344-9. [Crossref] [PubMed]
- Zhang YY, Mao HM, Wei CG, et al. Development and Validation of a Biparametric MRI Deep Learning Radiomics Model with Clinical Characteristics for Predicting Perineural Invasion in Patients with Prostate Cancer. Acad Radiol 2024;31:5054-65. [Crossref] [PubMed]



