
Nomogram to predict late extraluminal postpancreatectomy hemorrhage in patients with postoperative pancreatic fistula after pancreaticoduodenectomy
Highlight box
Key findings
• A nomogram was constructed to predict the risk of late extraluminal postpancreatectomy hemorrhage (LEPPH) in patients with postoperative pancreatic fistula after pancreaticoduodenectomy (PD).
What is known and what is new?
• Current research about prediction of LEPPH focuses merely on single aspect, resulting in poor generalizability and lacks a scale that can quantify bleeding risk.
• This study combines postoperative infection indicators with imaging examinations to make this nomogram more reliable and convenient to use.
What is the implication and what should change now?
• This nomogram could aid surgeons in early identification of patients prone to LEPPH following PD, enabling timely interventions and improving patient survival.
Introduction
Pancreaticoduodenectomy (PD) is currently the preferred surgical procedure for radical treatment of tumors in the pancreatic head, duodenum, and periampullary region. It is characterized by its complexity, significant trauma, high incidence of postoperative complications, and elevated mortality rates. Extraluminal postpancreatectomy hemorrhage (EPPH), although relatively uncommon among surgical complications, constitutes a crucial factor contributing to the overall postoperative mortality rate, ranging from 2.5% to 11.1% (1-3). Previous literature reviews have reported a mortality rate as high as 24.2–50% associated with postoperative EPPH (4,5).
The occurrence of EPPH can be classified into two phases: early and late (6). Early EPPH (EEPPH), manifesting within 24 hours post-surgery, is typically linked to inadequate vessel or tissue ligation during the surgical procedure or insufficient inspection of hemostatic conditions in the operative field before concluding the operation. It is widely regarded as a preventable complication. On the other hand, late EPPH (LEPPH), occurring 24 hours after surgery, involves bleeding outside the gastrointestinal tract within the abdominal cavity. This complication is often associated with ruptured intra-abdominal vessels or organ bleeding, and it is characterized by a sudden onset, rapid progression, and emerges as a major contributor to poor postoperative outcomes and increased mortality rates, particularly prevalent in patients developing pancreatic fistula following PD (7,8). Regarding the severity of LEPPH, it can be categorized into Grades A, B, and C. While Grade A requires no specific intervention, Grades B and C pose a significant threat to patients’ lives, necessitating early detection and treatment.
Recently, Dai et al. found that early removal of drainage may decrease incidence of complications after PD but not including hemorrhage (9). Some pancreatic features including gland texture and main pancreatic duct diameter (as risk factors of pancreatic fistula) were also included as variable in some studies focusing on PPH. Gland texture was not a risk factor of PPH (10). And Lu et al. found that pancreatic duct diameter contributed to late PPH (11). To better detect pancreatic duct diameter, Huscher et al. described a novel and safe intraoperative intraductal ultrasonography of the main pancreatic duct (12). Current predictors for LEPPH postoperatively primarily include general preoperative patient conditions, postoperative infection status, and postoperative abdominal computed tomography (CT) scans. Several studies suggested that preoperative indicators such as serum albumin concentration, total bilirubin levels, and preoperative jaundice treatment have poor accuracy in predicting LEPPH (13,14). Factors like abdominal infection have been identified as crucial contributors in some research (4,15-17). But existing observation indicators, such as postoperative fever, elevated white blood cell count, or positive drainage fluid cultures, are limited by their singular nature and subjective judgment, resulting in poor generalizability. Palumbo et al. chose postoperative imaging features, such as abdominal fluid distribution, bubble sign and anastomotic continuity, as predictive indicators (18). However, it only analyzes and predicts based on imaging characteristics, without combining other clinical characteristics of patients, and lacks a scale that can quantify bleeding risk, which makes its application less convenient and reliable.
We previously proposed a predictive model that combines postoperative infection indicators with imaging examinations (19). Findings indicated that continuously elevated neutrophil percentages on the third postoperative day, positive cultures in abdominal drainage fluid, fluid accumulation behind the pancreaticojejunostomy, and the presence of gas bubbles in the operative area independently contributed to the risk of post-pancreatic fistula bleeding. To further enhance predictive capabilities, we constructed a nomogram based on retrospective cases from West China Hospital, validating its predictive value in an external cohort of PD patients at three major pancreatic centers: West China Hospital, Sichuan Provincial People’s Hospital and Union Hospital, Tongji Medical College, Huazhong University of Science and Technology. We present this article in accordance with the STROBE reporting checklist (available at https://gs.amegroups.com/article/view/10.21037/gs-24-412/rc).
Methods
Patients
Flow chart of study design is shown in Figure 1.

For retrospective cases, inclusion criteria were: (I) age ≥18 years; (II) underwent PD; and (III) occurrence of postoperative pancreatic fistula (POPF). Exclusion criteria were: (I) EPPH within 24 hours postoperatively; (II) isolated gastrointestinal bleeding, such as from gastrointestinal ulcers or anastomotic site bleeding; (III) PPH happened before POPF; (IV) postoperative cardiovascular or cerebrovascular accidents, or pulmonary embolism; and (V) lack of postoperative imaging or missing more than 50% of the included indicators.
For prospective cases, inclusion criteria were: (I) age ≥18 years; (II) underwent PD; (III) occurrence of POPF; and (IV) voluntary participation with signed informed consent from patients’ family members or legal representatives. Exclusion criteria were: (I) EPPH within 24 hours postoperatively; (II) isolated gastrointestinal bleeding; (III) PPH happened before POPF; (IV) postoperative cardiovascular or cerebrovascular accidents, or pulmonary embolism; and (V) patients requesting withdrawal from the experimental study midway.
Based on the testing of the modeling data, it was estimated that a sample size of more than 200 cases in the external cohort would provide accurate validation results when the sample size of it reached approximately one-third of the modeling dataset.
The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). This study was approved by the Ethics Committee of West China Hospital, Sichuan University [approval No. 2022 Ethical Review (804); approval No. 2023 Ethical Review (2040)], and registered on the Chinese Clinical Trial Registry (ChiCTR2200065046 and ChiCTR2300077670). All participating hospitals were informed and agreed this study. Individual consent for retrospective cases was waived. Informed consent was obtained from all prospective patients.
Definitions
According to the 2016 International Study Group of Pancreatic Fistula (ISGPS), POPF is diagnosed when any measurable volume of drain fluid on or after postoperative day 3 with amylase level >3 times the upper limit, and is clinically relevant (20).
In accordance with the 2007 ISGPS, LEPPH refers to bleeding occurring after 24 hours postoperatively outside the digestive tract and within the abdominal cavity. It is categorized into mild, moderate, and severe based on clinical symptoms and the degree of hemoglobin decline (21).
As per the updated 2022 ISGPS definition, postoperative acute pancreatitis (AP) is considered when a patient exhibits: (I) persistently elevated postoperative serum amylase levels above the upper limit; (II) relevant clinical manifestations; and (III) imaging evidence of changes associated with AP (22).
Pancreaticoenteric anastomotic fluid accumulation was defined when postoperative imaging suggested fluid accumulation in the hepatic hilum, pancreaticoenteric anastomosis site on the ventral or dorsal aspect (Figure 2A).

The presence of bubbles in the abdominal fluid near the operative site is considered a positive bubble sign (Figure 2A). If bubbles are found near the drainage tube, consideration should be given to the possibility that the appearance of these bubbles is related to the entry of air from the drainage tube into the patient’s body, and a diagnosis should not be made (Figure 2B).
Postoperative imaging indicates discontinuous changes at the pancreaticoenteric anastomosis. If the discontinuous change is wedge-shaped and the maximum diameter is less than 2 mm, it is defined as a pancreaticoenteric anastomotic wedge fissure (Figure 2C).
If the diameter is greater than 2 mm, it is defined as cracking (Figure 2D).
All radiological features based on the CT scans 5 to 7 days after PD.
Statistical analysis
For data with a missing proportion not exceeding 10%, the “missForest” package in RStudio 4.3.1 software was used for multiple imputations, generating multiple imputed datasets, from which one dataset was randomly selected for subsequent analysis. Data with a missing proportion exceeding 10% were directly excluded. The “CompareGroups” package in RStudio 4.3.1 software was used for preprocessing the included data, employing Chi-squared or Fisher’s exact tests for categorical variables and t-tests or Wilcoxon rank-sum tests for continuous variables. P values <0.05 were considered statistically significant.
Variables with P values <0.05 in the analysis of bleeding and control groups were selected as candidate variables and included in the multivariate logistic regression model for multifactor analysis. Variables with P values <0.05 in the multifactor logistic regression model were finally included as risk factors in the prediction model. The retrospective participants from West China Hospital were randomly divided into a development cohort and an internal validation cohort at a ratio of “7:3”. In the development cohort, a logistic regression model served as the foundation, constructing a risk nomogram using the “rms” package in RStudio 4.3.1 software. The nomogram visualized the relationship between the cumulative scores of risk factors and the probability of occurrence. Subsequently, a receiver operating characteristic (ROC) curve was drawn, and the area under the ROC curve (AUC) was calculated to evaluate the model’s discrimination. Bootstrap resampling was performed 1,000 times for internal validation, and a calibration curve was drawn to assess the model’s calibration. The internal validation cohort was used to verify the stability of the predictive model by comparing the calibration curve trend with that of the development cohort. Finally, the calibration curve of the external data validation cohort was compared with those of the development cohort and the internal validation cohort to assess the generalization of the predictive model.
Results
Baseline characteristics of patients
A total of 2,924 retrospective cases underwent PD, and 695 patients (23.8%) experienced POPF, including 75 cases (2.6%) with LEPPH. According to inclusion criteria, a total of 420 retrospective cases were included, including 371 patients from West China Hospital (September 2011 to June 2020), 9 from Sichuan Provincial People’s Hospital and 40 from Wuhan Union Hospital (January 2021 to May 2022). A total of 467 prospective cases underwent PD, and 146 of them (31.3%) experienced POPF, including 23 cases (4.9%) with LEPPH. According to inclusion criteria, 131 patients were enrolled from June 2022 to July 2023, comprising 71 cases from West China Hospital, 10 from Provincial People’s Hospital, and 50 from Union Hospital.
Overall, 371 retrospective cases from West China Hospital were included and randomly divided into a development set of 259 cases and an internal validation set of 112 cases according to a ratio of 7:3. A total of 180 patients were eventually included as an external validation set. There were 71 prospective cases in West China Hospital, 19 in Sichuan Provincial People’s Hospital (9 retrospective and 10 prospective) and 90 in Wuhan Union Hospital (40 retrospective and 50 prospective).
Baseline data for 371 retrospective patients of development cohort and internal validation cohort are presented in Table 1. There were no significant differences in gender, age, BMI, smoking and drinking history between the two groups. The bleeding group had significantly longer hospital stays (21 vs. 30 days, P<0.001) and higher mortality (0.65% vs. 47.6%).
Table 1
| Variables | Controlled group (n=308) | LEPPH group (n=63) | P |
|---|---|---|---|
| Gender | 0.13 | ||
| Male | 127 (41.2) | 19 (30.2) | |
| Female | 181 (58.8) | 44 (69.8) | |
| Age (years) | 59.0 (50.8, 68.0) | 59.0 (52.0, 64.0) | 0.67 |
| BMI (kg/m2) | 23.1±3.21 | 23.1±2.72 | 0.92 |
| Smoking history | 103 (33.4) | 29 (46.0) | 0.08 |
| Drinking history | 83 (26.9) | 20 (31.7) | 0.54 |
| Diabetes | 21 (6.82) | 3 (4.76) | 0.78 |
| ASA score (≥3) | 56 (18.2) | 19 (30.2) | 0.047 |
| PBD | 39 (12.7) | 12 (19.0) | 0.25 |
| Preoperative TB (μmol/L) | 31.4 (11.1, 151) | 41.5 (13.8, 183) | 0.28 |
| Preoperative DB (μmol/L) | 22.8 (4.00, 131) | 35.5 (4.50, 170) | 0.31 |
| Preoperative albumin (g/L) | 40.5±5.58 | 40.6±5.74 | 0.90 |
| Clinical outcome | <0.001 | ||
| Recovery | 306 (99.4) | 33 (52.4) | |
| Mortality | 2 (0.65) | 30 (47.6) | |
| Duration of hospitalization (days) | 21.0 (16.0, 31.0) | 30.0 (23.0, 43.0) | <0.001 |
Data are presented as n (%), median (Q1, Q3) or mean ± standard deviation. ASA, American Society of Anesthesiologist; BMI, body mass index; DB, direct bilirubin; LEPPH, late extraluminal postpancreatectomy hemorrhage; PBD, preoperative biliary drainage; TB, total bilirubin.
Among all 98 cases of bleeding patients from both retrospective and prospective sets, the average time of first occurrence of LEPPH after surgery was 14.7 days. The bleeding locations were categorized into 36 cases with bleeding from rupture of the hepatic artery and its branches (36.73%), 20 cases involving bleeding at the pancreaticoenteric anastomosis site and surrounding vessels (20.41%), 19 cases with bleeding from rupture of the gastroduodenal artery stump (19.39%), 10 cases occurring in other intra-abdominal locations (10.2%), 8 cases (8.16%) where the specific bleeding site could not be promptly identified (these patients passed away soon after the LEPPH was detected and have no time to underwent examination to identify the specific bleeding sites), and 5 cases from the superior mesenteric artery (5.1%). Among these, 28 patients experienced moderate bleeding (28.57%), while the remaining 70 patients had severe bleeding (71.43%). Clinical outcomes included 60 patients who recovered and 38 patients who died.
Risk factors of LEPPH
Based on the data of patients in the bleeding group and the control group, the results of univariate analysis are presented in Table 2. Univariate analysis revealed differences between the two groups in variables such as pancreaticoenteric anastomotic dorsal fluid accumulation, bubble sign, pancreaticoenteric anastomotic cracking (PEAC), continuous elevation of neutrophil percentage after surgery, positive culture in intra-abdominal drainage fluid, postoperative AP, and sepsis. Subsequently, variables with statistically significant differences in univariate analysis were brought into multivariate analysis to obtain independent risk factors for postoperative bleeding. Pancreaticoenteric anastomotic dorsal fluid accumulation [odds ratio (OR) =7.873, 95% confidence interval (CI): 2.3–26.955, P=0.001], bubble sign (OR =7.542, 95% CI: 2.858–19.902, P<0.001), PEAC (OR =3.563, 95% CI: 1.073–11.837, P=0.04), surgery-related AP (OR =5.183, 95% CI: 2.038–13.182, P=0.001), and positive culture in intra-abdominal drainage fluid (OR =9.536, 95% CI: 3.351–27.133, P<0.001)—these five indicators showed significant statistical differences in multivariate logistic regression analysis (Table 2).
Table 2
| Variables | Controlled group (n=308) | LEPPH group (n=63) | Univariable logistic regression (P) | Multivariable logistic regression | ||
|---|---|---|---|---|---|---|
| OR (95% CI) | P | |||||
| Intraoperative | ||||||
| Blood loss (mL) | 200 [100, 400] | 300 [138, 500] | 0.44 | – | – | |
| Vascular reconstruction | 24 (7.79) | 2 (3.17) | 0.28 | – | – | |
| Postoperative | ||||||
| PEAFA | 84 (27.3) | 20 (31.7) | 0.26 | – | – | |
| PEADFA | 84 (27.3) | 51 (81.0) | <0.001 | 7.873 (2.3–26.955) | 0.001 | |
| Bubble sign | 39 (12.7) | 42 (66.7) | <0.001 | 7.542 (2.858–19.902) | <0.001 | |
| PEAC | 16 (5.19) | 15 (23.8) | <0.001 | 3.563 (1.073–11.837) | 0.04 | |
| Peak amylase (IU/L)* | 14,268 (37,334) | 18,756 (27,873) | 0.28 | – | – | |
| Minimum albumin (g/L) | 33.5 [29.9, 36.8] | 31.7 [29.0, 35.1] | 0.01 | – | – | |
| Persistent elevation of WBC | 206 (66.9) | 48 (76.2) | 0.30 | – | – | |
| Persistent elevation of Ne% | 91 (29.5) | 39 (61.9) | <0.001 | 2.007 (0.806–4.998) | 0.14 | |
| Positive culture* | 81 (26.3) | 54 (85.7) | <0.001 | 9.536 (3.351–27.133) | <0.001 | |
| Surgery-related AP | 50 (16.2) | 33 (52.4) | <0.001 | 5.183 (2.038–13.182) | 0.001 | |
| Sepsis | 11 (3.57) | 13 (20.6) | <0.001 | 2.769 (0.697–10.999) | 0.15 | |
| Bile leakage | 18 (5.84) | 8 (12.7) | 0.06 | – | – | |
Blood loss and minimum albumin are presented as median [Q1, Q3]; peak amylase is presented as median (interquartile range); other variables are presented as n (%). *, in drainage fluid. AP, acute pancreatitis; LEPPH, late extraluminal postpancreatectomy hemorrhage; Ne%, neutrophilic granulocyte percentage; PEAC, pancreaticoenteric anastomotic cracking; PEADFA, pancreaticoenteric anastomotic dorsal fluid accumulation; WBC, white blood cell.
Development and validation of a nomogram for predicting LEPPH
The statistically significant indicators in multivariable analyses—pancreaticoenteric anastomotic dorsal fluid accumulation, bubble sign, PEAC, surgery-related AP, and positive culture in intra-abdominal drainage fluid—were included in the predictive model.
A nomogram chart was created using data from the development cohort (Figure 3A). The scores of each variable are as follows: PEADFA [58], bubble sign [65], surgery-related AP [42], PEAC [53], positive culture [100]. The C-index of this nomogram was 0.932, and the calibration curve closely matched the ideal curve with a mean absolute error of 0.023 (Figure 3B).
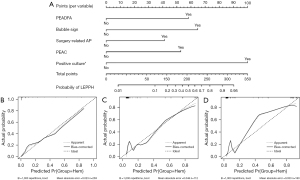
In the internal validation cohort, the calibration curve also exhibited a good fit with the ideal curve, with a C-index of 0.924 and mean absolute errors of 0.048 (Figure 3C).
External validation was conducted using 180 cases. A comparison with the development and internal validation dataset revealed heterogeneity in some indicators between the two datasets, helping to verify whether the risk prediction model had generalizability (Table 3). The calibration curve in the external validation set closely matched the ideal curve, with a C-index of 0.954 and mean absolute errors of 0.033 (Figure 3D).
Table 3
| Variables | External validation cases (n=180) | Retrospective cases (n=371) | P |
|---|---|---|---|
| Group | 0.56 | ||
| Controlled group | 145 (80.6) | 308 (83.0) | |
| LEPPH group | 35 (19.4) | 63 (17.0) | |
| Gender | 0.42 | ||
| Female | 78 (43.3) | 146 (39.4) | |
| Male | 102 (56.7) | 225 (60.6) | |
| Age (years) | 61.0 [52.0, 69.0] | 59.0 [51.0, 67.0] | 0.13 |
| BMI (kg/m2) | 23.1 (3.49) | 23.1 (3.13) | 0.83 |
| Smoking history | 47 (26.1) | 132 (35.6) | 0.03 |
| Drinking history | 34 (18.9) | 103 (27.8) | 0.03 |
| Diabetes | 17 (9.44) | 24 (6.47) | 0.28 |
| ASA score (≥3) | 23 (12.8) | 75 (20.2) | 0.043 |
| PBD | 54 (30.0) | 51 (13.7) | <0.001 |
| Blood loss (mL) | 200 [100, 400] | 200 [100, 400] | 0.12 |
| Vascular reconstruction | 13 (7.22) | 26 (7.01) | >0.99 |
| PEAFA | 72 (40.0) | 104 (28.0) | 0.01 |
| PEADFA | 45 (25.0) | 135 (36.4) | 0.006 |
| Bubble sign | 42 (23.3) | 81 (21.8) | 0.86 |
| Surgery related AP | 36 (20.0) | 83 (22.4) | 0.53 |
| PEAWF | 77 (42.8) | 92 (24.8) | <0.001 |
| PEAC | 32 (17.8) | 31 (8.36) | 0.002 |
| Peak amylase (IU/L)* | 13,865 (19,409) | 15,030 (35,910) | 0.62 |
| Positive culture* | 81 (45.0) | 135 (36.4) | 0.06 |
| Sepsis | 16 (8.89) | 24 (6.47) | 0.39 |
| Bile leakage | 17 (9.44) | 26 (7.01) | 0.41 |
| Persistent elevation of WBC | 83 (46.1) | 254 (68.5) | <0.001 |
| Persistent elevation of Ne% | 51 (28.3) | 130 (35.0) | 0.1 |
| Clinical outcome | 0.39 | ||
| Recovery | 169 (93.9) | 339 (91.4) | |
| Mortality | 11 (6.11) | 32 (8.63) | |
| Duration of hospitalization (days) | 22.0 [15.0, 30.0] | 23.0 [17.0, 33.0] | 0.13 |
Age, blood loss and duration of hospitalization are presented as median [Q1, Q3]; BMI is presented as mean (standard deviation); peak amylase is presented as median (interquartile range); other variables are presented as n (%). *, in drainage fluid. AP, acute pancreatitis; ASA, American Society of Anesthesiologist; BMI, body mass index; LEPPH, late extraluminal postpancreatectomy hemorrhage; Ne%, neutrophilic granulocyte percentage; PBD, preoperative biliary drainage; PEAC, pancreaticoenteric anastomotic cracking; PEADFA, pancreaticoenteric anastomotic dorsal fluid accumulation; PEAWF, pancreaticoenteric anastomotic wedge fissure; WBC, white blood cell.
Discussion
LEPPH, as one of the severe complications following PD, exhibits sudden onset, rapid progression, and high mortality rates (23). In this study, the mortality rate in the bleeding group (47.6%) was significantly higher than in the non-bleeding group (0.65%). Therefore, early identification and precise prediction of LEPPH can impede disease progression, ultimately improving patient prognosis.
The study constructed a nomogram capable of accurately predicting the occurrence of LEPPH in PD patients with POPF. Through internal and external validation, the nomogram demonstrated stability and generalizability. This tool enables surgeons to identify high-risk bleeding patients based on routine postoperative assessments, allowing for tailored preventive measures and increased success in rescue operations. For patients with POPF, we recommend performing a postoperative CT scan five days after surgery to evaluate the nomogram for the first time. If subsequent cultures yield positive results, the nomogram should be reevaluated.
Previous models for predicting LEPPH primarily focused on preoperative conditions and intraoperative surgical procedures (24-27). However, our research indicates that these factors are not stable predictors of LEPPH. For instance, it was conventionally believed that preoperative high total bilirubin levels were associated with LEPPH, and preoperative bilirubin reduction could lower bleeding risk by reducing serum total bilirubin levels (28). However, recent clinical studies suggest that patients receiving preoperative bilirubin reduction do not significantly reduce the risk of LEPPH, aligning with the results of this study (29-31). This predictive model offers a new perspective for surgeons managing PD postoperatively, allowing them to actively assess bleeding risk through postoperative examinations and implement timely measures. According to the predicted likelihood of LEPPH, we recommend that patients with a probability of less than 50% be transferred to a subordinate hospital for rehabilitation. Conversely, patients with a probability exceeding 50% should maintain a hemoglobin level above 80 g/L and remain under health surveillance in the surgical department for two to three weeks before being transferred to a subordinate hospital. Additionally, it provides a basis for future prospective trials for patients at high risk of bleeding after PD.
To construct the Nomogram, five variables were ultimately included in the predictive model: pancreaticoenteric anastomotic dorsal fluid accumulation, bubble sign, PEAC, surgery-related AP, and positive culture in intra-abdominal drainage fluid. These variables are routine postoperative examination items, making the model convenient for clinical use.
Pancreaticoenteric anastomotic dorsal fluid accumulation is related to poor healing at the dorsal aspect of the pancreaticoenteric anastomosis, leading to pancreatic fluid leakage and accumulation in the retroperitoneal space. Due to the anatomical characteristics of the retroperitoneal space, fluid containing high concentrations of digestive enzymes is challenging to drain effectively. As a result, long-term erosion of vessels around the anastomosis, including the residual end of the gastroduodenal artery and branches of the hepatic artery, can occur, eventually leading to rupture and bleeding (5).
The occurrence of the bubble sign has multiple causes. During the early data collection phase, gas bubbles inside the abdominal drainage tube were excluded from contrast-enhanced abdominal CT (Figure 2D). In the actual progression of the disease, the bubble sign can develop in two ways. Firstly, when the bubble sign is mainly distributed around the pancreaticoenteric anastomosis, it suggests poor healing at this site, with intestinal gas leaking to the surroundings of the anastomosis or other distant areas within the abdominal cavity. Simultaneously, the occurrence of the bubble sign may be related to the presence of gas-producing bacteria in intra-abdominal fluid, indicating a possible abdominal infection in patients (32).
PEAC is a less common imaging feature. Poor healing of pancreato-intestinal anastomosis may lead to serious pancreatic fistula and intestinal bacterial leakage contaminating the abdominal cavity, while aerogenic bacteria in the peritoneal effusion will cause continuous erosion of blood vessels of the operation sites, and eventually induce intraperitoneal vascular rupture and bleeding. When a clear discontinuity is observed in the pancreaticoenteric anastomosis on imaging, it alerts surgeons that the patient may have severe pancreatic fistula and intestinal fistula, raising concerns about serious intra-abdominal infection.
These three indicators are imaging features that surgeons often overlook. However, they hold significant clinical predictive value for the risk of LEPPH, emphasizing the importance of postoperative contrast-enhanced abdominal CT in assessing the recovery of the surgical area.
Postoperative AP is one of the more severe complications following PD, with its latest definition updated in 2022 by the ISGPF group (33). Limited research has been conducted on postoperative AP. Some clinical studies suggest a high correlation between postoperative AP and adverse events such as biliary fistula, pancreatic fistula, and organ failure (34). The specific underlying mechanisms remain unclear. In this study, the correlation between postoperative AP and LEPPH is identified as one of its unique predictive factors. In practical clinical settings, healthcare workers have not given sufficient attention to postoperative pancreatitis.
Positive culture in intra-abdominal drainage fluid is widely acknowledged as one of the predictive factors for bleeding, with abundant clinical research supporting this (35-38). Some studies suggested that when patients with pancreatic fistula have concomitant intra-abdominal infection, the pancreaticoenteric anastomosis underwent tissue edema and increased exudation under inflammatory stimulation. This further aggravates the severity of pancreatic fistula, triggering vascular and tissue bleeding. Simultaneously, metabolites of microorganisms may have erosive properties, further increasing the risk of bleeding in patients (39).
The Nomogram demonstrated good reliability (C-index =0.932) and exhibited good stability in the internal validation set and good generalizability in the external validation set. However, the external validation calibration curve showed “overestimation” of bleeding risk in certain intervals. This could be related to differences in the positive detection rate of risk factors between datasets. Comparing data differences between the development group and the external validation group revealed a significant statistical difference in two radiological indicators—pancreaticoenteric anastomotic dorsal fluid accumulation and PEAC. This may lead to bias in the final model’s predictive results compared to actual situations. Because prospective studies include postoperative contrast-enhanced CT as a mandatory item, while retrospective cases have some patients undergoing abdominal plain CT postoperatively. Previous studies have demonstrated that contrast-enhanced CT has significant advantages in detecting and locating fluid around the pancreas, evaluating pancreatic tissue exudation, and assessing pancreatic parenchymal lesions (31,40-43).
Conclusions
The nomogram, constructed based on a combination of pancreaticoenteric anastomotic dorsal fluid accumulation, bubble sign, PEAC, postoperative AP, and positive culture in intra-abdominal drainage fluid, exhibited excellent predictive capabilities for LEPPH after PD. It could aid surgeons in early identification of patients prone to LEPPH following PD, enabling timely interventions and improving patient survival.
Acknowledgments
None.
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at https://gs.amegroups.com/article/view/10.21037/gs-24-412/rc
Data Sharing Statement: Available at https://gs.amegroups.com/article/view/10.21037/gs-24-412/dss
Peer Review File: Available at https://gs.amegroups.com/article/view/10.21037/gs-24-412/prf
Funding: This work was supported by
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://gs.amegroups.com/article/view/10.21037/gs-24-412/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). This study was approved by the Ethics Committee of West China Hospital, Sichuan University [approval No. 2022 Ethical Review (804); approval No. 2023 Ethical Review (2040)], and registered on the Chinese Clinical Trial Registry (ChiCTR2200065046 and ChiCTR2300077670). All participating hospitals were informed and agreed the study. Individual consent for retrospective cases was waived. Informed consent was obtained from all prospective patients.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Schnelldorfer T, Adams DB, Warshaw AL, et al. Forgotten pioneers of pancreatic surgery: beyond the favorite few. Ann Surg 2008;247:191-202. [Crossref] [PubMed]
- Yang YM, Tian XD, Zhuang Y, et al. Risk factors of pancreatic leakage after pancreaticoduodenectomy. World J Gastroenterol 2005;11:2456-61. [Crossref] [PubMed]
- Chen XP, Huang ZY, Lau JW, et al. Chen's U-suture technique for end-to-end invaginated pancreaticojejunostomy following pancreaticoduodenectomy. Ann Surg Oncol 2014;21:4336-41. [Crossref] [PubMed]
- Yeh TS, Jan YY, Jeng LB, et al. Pancreaticojejunal anastomotic leak after pancreaticoduodenectomy--multivariate analysis of perioperative risk factors. J Surg Res 1997;67:119-25. [Crossref] [PubMed]
- Piao S, Pan Z, Qian C, et al. The effect of bilateral U-sutures in pancreaticojejunostomy in 75 consecutive cases. Acta Chir Belg 2019;119:201-4. [Crossref] [PubMed]
- Marcus SG, Cohen H, Ranson JH. Optimal management of the pancreatic remnant after pancreaticoduodenectomy. Ann Surg 1995;221:635-45; discussion 645-8. [Crossref] [PubMed]
- Grobmyer SR, Kooby D, Blumgart LH, et al. Novel pancreaticojejunostomy with a low rate of anastomotic failure-related complications. J Am Coll Surg 2010;210:54-9. [Crossref] [PubMed]
- Papachristou DN, Fortner JG. Pancreatic fistula complicating pancreatectomy for malignant disease. Br J Surg 1981;68:238-40. [Crossref] [PubMed]
- Dai M, Liu Q, Xing C, et al. Early drain removal after major pancreatectomy reduces postoperative complications: a single-center, randomized, controlled trial. Journal of Pancreatology 2020;3:93-100.
- Duan Y, Du Y, Mu Y, et al. Development and validation of a novel predictive model for postpancreatectomy hemorrhage using lasso-logistic regression: an international multicenter observational study of 9631 pancreatectomy patients. Int J Surg 2025;111:791-806. [Crossref] [PubMed]
- Lu JW, Ding HF, Wu XN, et al. Intra-abdominal hemorrhage following 739 consecutive pancreaticoduodenectomy: Risk factors and treatments. J Gastroenterol Hepatol 2019;34:1100-7. [Crossref] [PubMed]
- Huscher CGS, Lazzarin G, Marchegiani G, et al. Intraoperative intraductal ultrasonography of the main pancreatic duct during pancreatoduodenectomy: technical description of a pilot series. Updates Surg 2024;76:2471-6. [Crossref] [PubMed]
- Osada S, Imai H, Sasaki Y, et al. Reconstruction method after pancreaticoduodenectomy. Idea to prevent serious complications. JOP 2012;13:1-6.
- Choe YM, Lee KY, Oh CA, et al. Risk factors affecting pancreatic fistulas after pancreaticoduodenectomy. World J Gastroenterol 2008;14:6970-4. [Crossref] [PubMed]
- Xiang Y, Wu J, Lin C, et al. Pancreatic reconstruction techniques after pancreaticoduodenectomy: a review of the literature. Expert Rev Gastroenterol Hepatol 2019;13:797-806. [Crossref] [PubMed]
- Berger AC, Howard TJ, Kennedy EP, et al. Does type of pancreaticojejunostomy after pancreaticoduodenectomy decrease rate of pancreatic fistula? A randomized, prospective, dual-institution trial. J Am Coll Surg 2009;208:738-47; discussion 747-9. [Crossref] [PubMed]
- Langrehr JM, Bahra M, Jacob D, et al. Prospective randomized comparison between a new mattress technique and Cattell (duct-to-mucosa) pancreaticojejunostomy for pancreatic resection. World J Surg 2005;29:1111-9, discussion 1120-1. [Crossref] [PubMed]
- Palumbo D, Tamburrino D, Partelli S, et al. Before sentinel bleeding: early prediction of postpancreatectomy hemorrhage (PPH) with a CT-based scoring system. Eur Radiol 2021;31:6879-88. [Crossref] [PubMed]
- Yang J, Li YC, Liu XB, et al. Infection and image findings to predict delayed hemorrhage in postoperative pancreatic fistula patients after pancreaticoduodenectomy. Asian J Surg 2022;45:1130-1. [Crossref] [PubMed]
- Bassi C, Marchegiani G, Dervenis C, et al. The 2016 update of the International Study Group (ISGPS) definition and grading of postoperative pancreatic fistula: 11 Years After. Surgery 2017;161:584-91. [Crossref] [PubMed]
- Wente MN, Veit JA, Bassi C, et al. Postpancreatectomy hemorrhage (PPH): an International Study Group of Pancreatic Surgery (ISGPS) definition. Surgery 2007;142:20-5. [Crossref] [PubMed]
- Marchegiani G, Barreto SG, Bannone E, et al. Postpancreatectomy Acute Pancreatitis (PPAP): Definition and Grading From the International Study Group for Pancreatic Surgery (ISGPS). Ann Surg 2022;275:663-72. [Crossref] [PubMed]
- Simon R. Complications After Pancreaticoduodenectomy. Surg Clin North Am 2021;101:865-74. [Crossref] [PubMed]
- Wellner UF, Kulemann B, Lapshyn H, et al. Postpancreatectomy hemorrhage--incidence, treatment, and risk factors in over 1,000 pancreatic resections. J Gastrointest Surg 2014;18:464-75. [Crossref] [PubMed]
- Chincarini M, Zamboni GA, Pozzi Mucelli R. Major pancreatic resections: normal postoperative findings and complications. Insights Imaging 2018;9:173-87. [Crossref] [PubMed]
- Ricci C, Casadei R, Buscemi S, et al. Late postpancreatectomy hemorrhage after pancreaticoduodenectomy: is it possible to recognize risk factors? JOP 2012;13:193-8.
- Pecorelli N, Carrara G, De Cobelli F, et al. Effect of sarcopenia and visceral obesity on mortality and pancreatic fistula following pancreatic cancer surgery. Br J Surg 2016;103:434-42. [Crossref] [PubMed]
- Shen Z, Zhang J, Zhao S, et al. Preoperative biliary drainage of severely obstructive jaundiced patients decreases overall postoperative complications after pancreaticoduodenectomy: A retrospective and propensity score-matched analysis. Pancreatology 2020;20:529-36. [Crossref] [PubMed]
- De Pastena M, Marchegiani G, Paiella S, et al. Impact of preoperative biliary drainage on postoperative outcome after pancreaticoduodenectomy: An analysis of 1500 consecutive cases. Dig Endosc 2018;30:777-84. [Crossref] [PubMed]
- Wang D, Lin H, Guan C, et al. Impact of preoperative biliary drainage on postoperative complications and prognosis after pancreaticoduodenectomy: A single-center retrospective cohort study. Front Oncol 2022;12:1037671. [Crossref] [PubMed]
- Amaral MJ, Freitas J, Amaral M, et al. Clinical Impact of Preoperative Biliary Drainage in Patients with Ductal Adenocarcinoma of the Pancreatic Head. Diagnostics (Basel) 2023;13:1281. [Crossref] [PubMed]
- Kilambi R, Singh AN. Duct-to-mucosa versus dunking techniques of pancreaticojejunostomy after pancreaticoduodenectomy: Do we need more trials? A systematic review and meta-analysis with trial sequential analysis. J Surg Oncol 2018;117:928-39. [Crossref] [PubMed]
- Daamen LA, Smits FJ, Besselink MG, et al. A web-based overview, systematic review and meta-analysis of pancreatic anastomosis techniques following pancreatoduodenectomy. HPB (Oxford) 2018;20:777-85. [Crossref] [PubMed]
- Zhang S, Lan Z, Zhang J, et al. Duct-to-mucosa versus invagination pancreaticojejunostomy after pancreaticoduodenectomy: a meta-analysis. Oncotarget 2017;8:46449-60. [Crossref] [PubMed]
- Sun X, Zhang Q, Zhang J, et al. Meta-analysis of invagination and duct-to-mucosa pancreaticojejunostomy after pancreaticoduodenectomy: An update. Int J Surg 2016;36:240-7. [Crossref] [PubMed]
- Hua J, He Z, Qian D, et al. Duct-to-Mucosa Versus Invagination Pancreaticojejunostomy Following Pancreaticoduodenectomy: a Systematic Review and Meta-Analysis. J Gastrointest Surg 2015;19:1900-9. [Crossref] [PubMed]
- Senda Y, Shimizu Y, Natsume S, et al. Randomized clinical trial of duct-to-mucosa versus invagination pancreaticojejunostomy after pancreatoduodenectomy. Br J Surg 2018;105:48-57. [Crossref] [PubMed]
- Singh AN, Pal S, Mangla V, et al. Pancreaticojejunostomy: Does the technique matter? A randomized trial. J Surg Oncol 2018;117:389-96. [Crossref] [PubMed]
- Bai X, Zhang Q, Gao S, et al. Duct-to-Mucosa vs Invagination for Pancreaticojejunostomy after Pancreaticoduodenectomy: A Prospective, Randomized Controlled Trial from a Single Surgeon. J Am Coll Surg 2016;222:10-8. [Crossref] [PubMed]
- Callery MP, Pratt WB, Kent TS, et al. A prospectively validated clinical risk score accurately predicts pancreatic fistula after pancreatoduodenectomy. J Am Coll Surg 2013;216:1-14. [Crossref] [PubMed]
- Seykora TF, Ecker BL, McMillan MT, et al. The Beneficial Effects of Minimizing Blood Loss in Pancreatoduodenectomy. Ann Surg 2019;270:147-57. [Crossref] [PubMed]
- Casciani F, Trudeau MT, Asbun HJ, et al. The effect of high intraoperative blood loss on pancreatic fistula development after pancreatoduodenectomy: An international, multi-institutional propensity score matched analysis. Surgery 2021;170:1195-204. [Crossref] [PubMed]
- Wolk S, Radosa CG, Distler M, et al. Risk Factors for In-hospital Mortality After Transarterial Intervention After Postpancreatectomy Hemorrhage. Cardiovasc Intervent Radiol 2020;43:1342-52. [Crossref] [PubMed]


