The role of noncoding RNA in thyroid cancer
Abstract: Increasing noncoding RNAs (ncRNAs) were found to show abnormal expression patterns in various human cancers. Based on size, ncRNAs are generally grouped into two categories, short noncoding RNAs and long noncoding RNAs (lncRNAs) of greater than 200 nt. Small noncoding RNAs include microRNAs, piRNAs, snoRNAs, and endogenous siRNAs, out of the role of miRNAs in development and cancer biology has been extensively studied. In contrast to small noncoding RNAs like miRNAs, long noncoding RNAs are much less known concerning their functions in human cancers especially in thyroid cancer. The present review highlighted the roles of miRNAs and newly discovered lncRNAs in thyroid development, tumorigenesis, metastasis, and their clinical implication.
Key words: MicroRNA; long noncoding RNA; thyroid cancer
Introduction
Less than 2% the mammalian genome are protein coding genes and over 90% of genome represent noncoding RNAs (ncRNA) which are transcribed but do not encode proteins. Despite of initial controversy regarding their biological characters, increasing evidence had showed that ncRNAs are highly regulated and functional (1). Two categories of ncRNA were grouped according to their size, short ncRNAs and long ncRNAs (lncRNA). The short ncRNA includes transcripts such as miRNAs, transfer RNAs (tRNAs), small interfering RNAs (siRNAs), piwi-interacting RNAs (piRNAs) and some ribosomal RNAs, out of which miRNAs were most extensively studied in human cancers. Although being not well understood in human cancers, long noncoding RNAs (>200 nt) have recently forwarded to the forefront of noncoding RNA research. Thyroid cancer is the most common malignant tumor of the endocrine organs whose incidence has been steadily increased over the past few decades (2). The deregulation of ncRNA expression is believed to be an important regulator of tumor development and progression of thyroid cancer. The present review highlighted the roles of miRNAs and newly discovered lncRNAs in thyroid development, tumorigenesis, metastasis, and their clinical implication.
MiRNAs and thyroid cancer
MicroRNAs (miRNAs) represent a class of short endogenous noncoding RNAs regulating gene expression at mRNA post-transcriptional level in many biological and pathological processes, including proliferation, apoptosis, and differentiation (3). Increasing evidence has revealed the involvement of mi-RNA in human malignancies. The deregulation of miRNA expression is believed to be an important regulator of tumor development and progression. Due to its repression effect, deregulation of specific mi-RNA could lead to the repression of tumor suppressor gene and/or increase of oncogene expression. Consequently, these molecular changes favor cell proliferation, differentiation and apoptosis. MicroRNA expression profiling of human tumors has identified signatures associated with diagnosis, staging, prognosis, and response to treatment (4). MiRNA expression profiles resulted in being different not only between tumors and normal tissues but also between different subtypes of tumors and between primary tumors and metastatic tumors.
A comprehensive analysis by microarray found that a significant miRNA signature with thyroid cancers. Out of numerous differentially expressed miRNAs in thyroid cancer, three miRNAs including miR-221, -222 and -181b were extensively studies in thyroid cancers (5). Studies demonstrated that miR-221, -222, and -146 are transcriptionally up-regulated in PTC tumors in comparison with normal thyroid tissue (6). Functional study showed that miR-221 and miR-222 are endogenous regulators of P27Kip1 protein expression, which represents a very important regulator of cell cycle (7). What is concomitant with up-regulation of the three miRNAs is the dramatic loss of KIT transcript and Kit protein, both of which involves in the pathogenesis of thyroid cancer. Microarray analysis of PTCs showed numerous genes were directly and indirectly regulated by miR-221 and studies both in vitro and in vivo using the bioluminescence imaging system confirmed the down-regulation of HOXB5 by endogenous or exogenous miR-221 (8). Significant down-regulation of miR-1 was detected in a panel of thyroid tumors compared with normal thyroid tissues and miR-1 as a tumor suppressor targeting CCND2, CXCR4, and SDF-1 genes, suggesting its ability to inhibit thyroid carcinoma cell proliferation and migration (9).
In contrast to mRNAs, mature miRNAs are comparatively stable and remain largely intact in routinely collected, formalin-fixed paraffin-embedded (FFPE) clinical tissues (10). The ability to detect miRNA profiles in FFPE tissues implicated a great opportunity to perform the large retrospective analyses necessary to confirm the diagnostic role and investigate the prognostic significance of miRNA profiles. Numerous studies have demonstrated that the potential diagnostic value of mi-RNA expression signatures in thyroid cancer, especially for indeterminate results on fine-needle aspiration biopsy (FNAB) samples. FNAB is currently the most widely used tool for the preoperative diagnosis of thyroid lesions with limitation for up to 30% indeterminate cases (11). Investigation of miRNA expression pattern for differential diagnosis of thyroid neoplasms in fine needle aspiration biopsy samples is feasible and may improve the accuracy of FNAB cytology. Pallante et al. (5) investigated that expression of miR-221, -222 and -181b had 5- to 35-fold differential in FNAB samples of PTCs compared with other thyroid nodules. Overexpression of four miRNAs (miR-100, miR-125b, miR-138, and miR-768-3p) was detected in malignant samples of follicular origin and only miR-125b was significantly overexpressed in FTC samples (12). These findings suggested that specific miRNAs can be potential diagnostic tools with high accuracy in both surgical and preoperative FNA samples. Mazeh et al. (13) found that miR-221 was the most favorable miRNA in differentiating benign from malignant thyroid pathology with specificity (100%), negative (96%) and positive (100%) predictive value, and accuracy (98%) respectively. Given the high negative predictive value of miR-7 (100%), patients may benefit from the result based on the predictor and avoid an immediate diagnostic thyroidectomy.
Mi-RNA expression profiles have a close association with clinicopathological features which help determine optimum management of thyroid cancer. A recent study suggested that miRNA signature can distinguishes the degree of PTC aggressiveness (14). The results showed that four miRNAs (miR-146b, miR-222, miR-34b, miR-130b) were differentially expressed in aggressive in comparison with nonaggressive PTCs. Additionally, miR-146b was demonstrated to have a close association with aggressive behavior of PTC among BRAF-positive tumors, which further refine the prognostic importance of BRAF. Similar correlation was observed between downregulated miRNAs (miR-34b and miR-1) and higher MET expression in aggressive PTC. Chou et al. (15) uncovered that overexpression of miR-146b, miR-221, and miR-222 were significantly associated with extra-thyroidal invasion in PTCS. MiRNA-100 was observed to have a significantly expression level between T1 and T4 tumors (16). Schwertheim et al. (17) reported that poorly differentiated thyroid carcinoma had a distinct mi-RNA expression profile in comparison with both PTC and ATC, suggesting that deregulation of some miRNAs may take part in selecting a subset of PTC progressing to PDTC.
LncRNAs and thyroid cancer
Increasing studies suggest that lncRNAs constitute an important component of tumor biology, representing regulatory functions including modulation of apoptosis and invasion, reprogramming of induced pluripotent stem cells, marker of cell fate and parental imprinting. Deregulation of individual lncRNA expression not only involve in development and progression of specific cancers, but also be significant molecules for clinical implication (18). LncRNAs utilize varied mechanism to regulate gene expression. In general, the mechanism lncRNA regulating gene expressing could be transcriptional or post-transcriptional. Cis- and trans-regulation are two main transcriptional regulations, under which lncRNAs can target genomically local and distant genes respectively. The post-transcriptional regulatory mechanism is involved in post-transcriptional processing of mRNAs, including splicing, editing, trafficking, translation and degradation. Recently, a new regulatory mechanism has been revealed that lncRNA can function as competing endogenous RNA (termed ceRNA) for shared miRNAs (19,20). ceRNAs showed a post-transcriptional regulatory role in miRNA molecules’ distribution on the targets. The schematic of lncRNAs was illustrated in Figure 1.
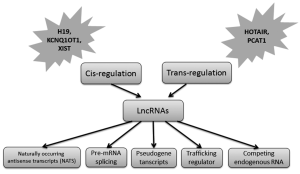
Out of numerous lncRNAs, a few well characterized lncRNAs to date such as HOTAIR, H19, MALAT1, Xist, KCNQ1OT1, AIR, and Evf-2 were extensively studied in human cancers. HOTAIR (HOX Antisense Intergenic RNA) can distally regulate the chromosomal domain on HOXD locus via physical interaction of its 5’ domain with PRC2 methylase and in turn result in silencing of the HOXD gene (21). HOTAIR is found to be significantly up-regulated in breast cancer and hepatocellular carcinoma and can be served as an independent predictor of prognosis (18,22). H19, one of the imprinting-associated lncRNAs, has been observed to be deregulated in hepatocellular and bladder cancer and involve in both oncogenic and tumor suppressive qualities. The diverse roles of H19 in cancers may be demonstrated due to its direct activation by cMYC and down-regulation by p53 (23,24). Abnormal expression of MALAT1 was investigated in various human cancers including breast, lung, pancreas, prostate, liver, colon, and ovarian cancer (25,26). Silencing of MALAT1 expression in lung cancer cells can impair cell migration ability by regulation of motility-related genes.
The role of lncRNAs in thyroid cancer is just beginning to be elucidated and there is a long way to go. Most recently, a genome-wide association studies (GWAS) addressed the predisposition to papillary thyroid cancer (PTC), out of which two SNPs (rs965513 and rs944289) were addressed to have highly significant association with PTC located in 9q22.33 and 14q13.3 respectively (27-30). A long noncoding RNA gene termed Papillary Thyroid Carcinoma Susceptibility Candidate 3 (PTCSC3) located 3.2 kb downstream of rs944289 at 14q.13.3 (31). PTCSC3 expression was found to be strictly thyroid-specific and be dramatically down-regulated in both thyroid tumor tissues and thyroid cell lines. However, the function of PTCSC3 in thyroid cancer is still unclear. Yoon et al. (32) reported that down-regulation of a novel gene, NAMA (noncoding RNA associated with MAP kinase pathway and growth arrest) was highly associated with the activating BRAF mutation V600E in papillary thyroid cancer.
Up to date, only a small portion of lncRNAs has been identified and the implication of lncRNAs in tumorigenesis, metastasis, and progression remain to be further investigated. LncRNAs have emerged as important regulatory molecules in development and progression of thyroid cancer. Given the expression profiling of lncRNAs in human cancers is not fully reported, study of lncRNAs in thyroid cancer is becoming an attractive field, which will lead to new markers of cancer diagnosis, prognosis as well as novel therapeutic targets.
Acknowledgements
Disclosure: The authors declare no conflict of interest.
References
- ENCODE Project Consortium, Birney E, Stamatoyannopoulos JA, et al. Identification and analysis of functional elements in 1% of the human genome by the ENCODE pilot project. Nature 2007;447:799-816.
- Davies L, Welch HG. Increasing incidence of thyroid cancer in the United States, 1973-2002. JAMA 2006;295:2164-7.
- Bartel DP. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 2004;116:281-97.
- Lu J, Getz G, Miska EA, et al. MicroRNA expression profiles classify human cancers. Nature 2005;435:834-8.
- Pallante P, Visone R, Ferracin M, et al. MicroRNA deregulation in human thyroid papillary carcinomas. Endocr Relat Cancer 2006;13:497-508.
- He H, Jazdzewski K, Li W, et al. The role of microRNA genes in papillary thyroid carcinoma. Proc Natl Acad Sci U S A 2005;102:19075-80. .
- Visone R, Russo L, Pallante P, et al. MicroRNAs (miR)-221 and miR-222, both overexpressed in human thyroid papillary carcinomas, regulate p27Kip1 protein levels and cell cycle. Endocr Relat Cancer 2007;14:791-8.
- Kim HJ, Kim YH, Lee DS, et al. In vivo imaging of functional targeting of miR-221 in papillary thyroid carcinoma. J Nucl Med 2008;49:1686-93.
- Leone V, D’Angelo D, Rubio I, et al. MiR-1 is a tumor suppressor in thyroid carcinogenesis targeting CCND2, CXCR4, and SDF-1alpha. J Clin Endocrinol Metab 2011;96:E1388-98.
- Tetzlaff MT, Liu A, Xu X, et al. Differential expression of miRNAs in papillary thyroid carcinoma compared to multinodular goiter using formalin fixed paraffin embedded tissues. Endocr Pathol 2007;18:163-73.
- American Thyroid Association (ATA) Guidelines Taskforce on Thyroid Nodules and Differentiated Thyroid Cancer, Cooper DS, Doherty GM, et al. Revised American Thyroid Association management guidelines for patients with thyroid nodules and differentiated thyroid cancer. Thyroid 2009;19:1167-214.
- Vriens MR, Weng J, Suh I, et al. MicroRNA expression profiling is a potential diagnostic tool for thyroid cancer. Cancer 2012;118:3426-32.
- Mazeh H, Mizrahi I, Halle D, et al. Development of a microRNA-based molecular assay for the detection of papillary thyroid carcinoma in aspiration biopsy samples. Thyroid 2011;21:111-8.
- Yip L, Kelly L, Shuai Y, et al. MicroRNA signature distinguishes the degree of aggressiveness of papillary thyroid carcinoma. Ann Surg Oncol 2011;18:2035-41.
- Chou CK, Chen RF, Chou FF, et al. miR-146b is highly expressed in adult papillary thyroid carcinomas with high risk features including extrathyroidal invasion and the BRAF(V600E) mutation. Thyroid 2010;20:489-94.
- Vriens MR, Weng J, Suh I, et al. MicroRNA expression profiling is a potential diagnostic tool for thyroid cancer. Cancer 2012;118:3426-32.
- Schwertheim S, Sheu SY, Worm K, et al. Analysis of deregulated miRNAs is helpful to distinguish poorly differentiated thyroid carcinoma from papillary thyroid carcinoma. Horm Metab Res 2009;41:475-81.
- Gupta RA, Shah N, Wang KC, et al. Long non-coding RNA HOTAIR reprograms chromatin state to promote cancer metastasis. Nature 2010;464:1071-6.
- Salmena L, Poliseno L, Tay Y, et al. A ceRNA hypothesis: the Rosetta Stone of a hidden RNA language? Cell 2011;146:353-8.
- McCarthy N. Regulatory RNA: layer by layer. Nat Rev Genet 2011;12:804.
- Rinn JL, Kertesz M, Wang JK, et al. Functional demarcation of active and silent chromatin domains in human HOX loci by noncoding RNAs. Cell 2007;129:1311-23.
- Gibb EA, Brown CJ, Lam WL. The functional role of long non-coding RNA in human carcinomas. Mol Cancer 2011;10:38.
- Barsyte-Lovejoy D, Lau SK, Boutros PC, et al. The c-Myc oncogene directly induces the H19 noncoding RNA by allele-specific binding to potentiate tumorigenesis. Cancer Res 2006;66:5330-7.
- Lee MP, DeBaun MR, Mitsuya K, et al. Loss of imprinting of a paternally expressed transcript, with antisense orientation to KVLQT1, occurs frequently in Beckwith-Wiedemann syndrome and is independent of insulin-like growth factor II imprinting. Proc Natl Acad Sci U S A 1999;96:5203-8.
- Lai MC, Yang Z, Zhou L, et al. Long non-coding RNA MALAT-1 overexpression predicts tumor recurrence of hepatocellular carcinoma after liver transplantation. Med Oncol 2012;29:1810-6.
- Lin R, Maeda S, Liu C, et al. A large noncoding RNA is a marker for murine hepatocellular carcinomas and a spectrum of human carcinomas. Oncogene 2007;26:851-8.
- Gudmundsson J, Sulem P, Gudbjartsson DF, et al. Common variants on 9q22.33 and 14q13.3 predispose to thyroid cancer in European populations. Nat Genet 2009;41:460-4.
- Gudmundsson J, Sulem P, Gudbjartsson DF, et al. Discovery of common variants associated with low TSH levels and thyroid cancer risk. Nat Genet 2012;44:319-22.
- Takahashi M, Saenko VA, Rogounovitch TI, et al. The FOXE1 locus is a major genetic determinant for radiation-related thyroid carcinoma in Chernobyl. Hum Mol Genet 2010;19:2516-23.
- He H, Nagy R, Liyanarachchi S, et al. A susceptibility locus for papillary thyroid carcinoma on chromosome 8q24. Cancer Res 2009;69:625-31.
- Jendrzejewski J, He H, Radomska HS, et al. The polymorphism rs944289 predisposes to papillary thyroid carcinoma through a large intergenic noncoding RNA gene of tumor suppressor type. Proc Natl Acad Sci U S A 2012;109:8646-51.
- Yoon H, He H, Nagy R, et al. Identification of a novel noncoding RNA gene, NAMA, that is downregulated in papillary thyroid carcinoma with BRAF mutation and associated with growth arrest. Int J Cancer 2007;121:767-75.


